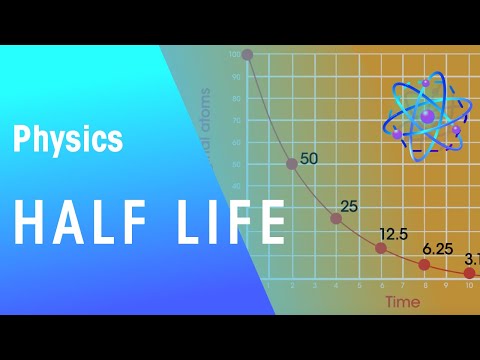
In general there is an inverse relation between the half-life and the intensity of radioactivity of an isotope. Isotopes with a long half-life decay very slowly, and so produce fewer radioactive decays per second; their intensity is less. Istopes with shorter half-lives are more intense.
Q. What is the half life of a radioisotope If a 50g sample becomes 25g after 18 days?
Answer: 18 DAYS IS THE HALF LIFE OF THE RADIOISOTOPE.
Table of Contents
Q. What does a drug’s half life mean?
What is a drug’s half-life? The half-life of a drug is the time it takes for the amount of a drug’s active substance in your body to reduce by half. This depends on how the body processes and gets rid of the drug. It can vary from a few hours to a few days, or sometimes weeks.
Q. What is the half life symbol called?
λ
Q. How many hours should be between medications?
Try to divide up your dosing times as evenly as possible throughout the day: for example, every 12 hours for a drug that needs to be taken twice a day, or every 8 hours for a drug that needs to be taken three times a day.
Q. What is the safest element?
Thorium is a weakly radioactive metallic chemical element with the symbol Th and atomic number 90….
| Thorium | |
|---|---|
| Appearance | silvery, often with black tarnish |
| Standard atomic weight Ar, std(Th) | 232.0377(4) |
| Thorium in the periodic table | |
Q. How harmful is plutonium?
Because it emits alpha particles, plutonium is most dangerous when inhaled. When plutonium particles are inhaled, they lodge in the lung tissue. The alpha particles can kill lung cells, which causes scarring of the lungs, leading to further lung disease and cancer.
Q. Why is plutonium so rare?
The reason that plutonium (and other transuranic elements) are so rare in nature is that being radioactive, they decay with a characteristic half-life. Any element formed at that time with a half-life much less than the Earth’s age–or 4.5 billion year–has nearly all decayed into lighter elements by now.